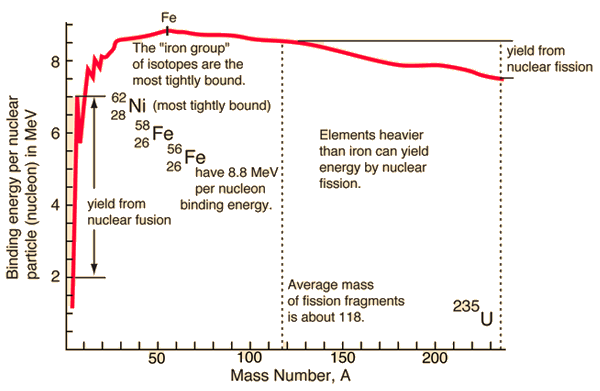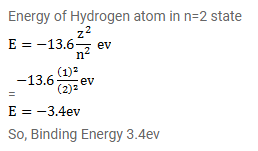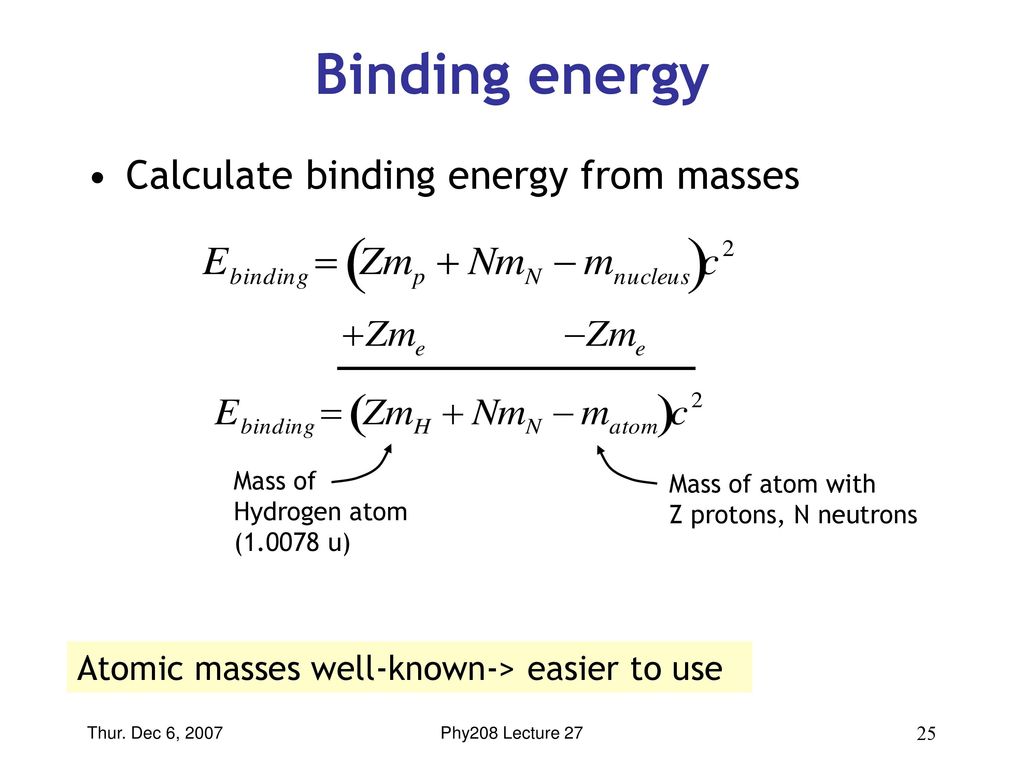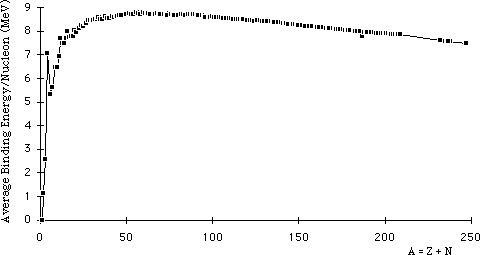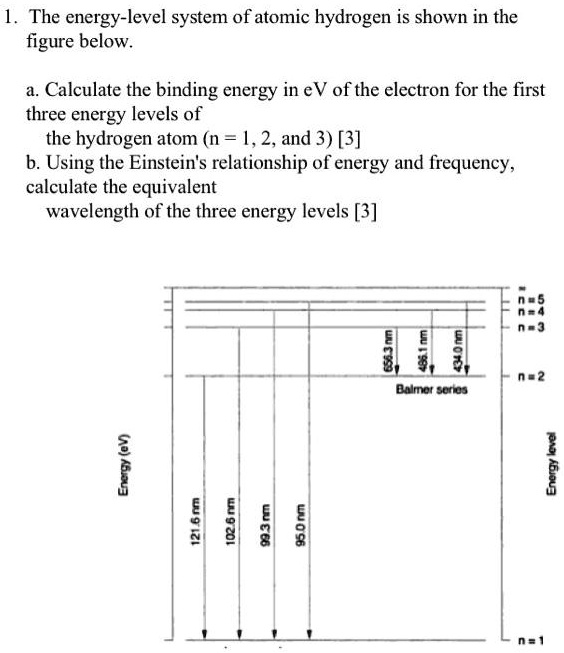
SOLVED: The energy-level system of atomic hydrogen is shown in the figure below. Calculate the binding energy in eV of the electron for the first three energy levels of the hydrogen atom (

If the binding energy of the electron in hydrogen atom is 13 6 eV, calculate the energy required to remove - Physics - Atoms - 12597845 | Meritnation.com
If the binding energy of the electron in a hydrogen atom is 13.6 eV, - Sarthaks eConnect | Largest Online Education Community

In a hydrogen atom,the binding energy of the electron in the n^th state is En , then the frequency of revolution of the electron in the nth orbits is

The binding energy of `e^(-)` in ground state of hydrogen atom is 13.6 eV . The energies required to - YouTube
97 binding energy of electrons in ground state of Hatomis 13.6eV ,energies required to eject out an electrons from three lowest states of He + atom willl be 1)13.6,10.2,3.4 2)13.6,3.4,1.5 3)13.6,27.2,40.8 4)54.5,13.6,6 explain??

A hydrogen atom in a state having a binding energy of 0.85 eV makes transition to a state with - YouTube
If the binding energy of the electron in the ground state of hydrogen atom is E, then the frequency of electron in the nth orbit is

In a hydrogen atom, the binding energy of the electron in the ground state is `E_(1)` then - YouTube